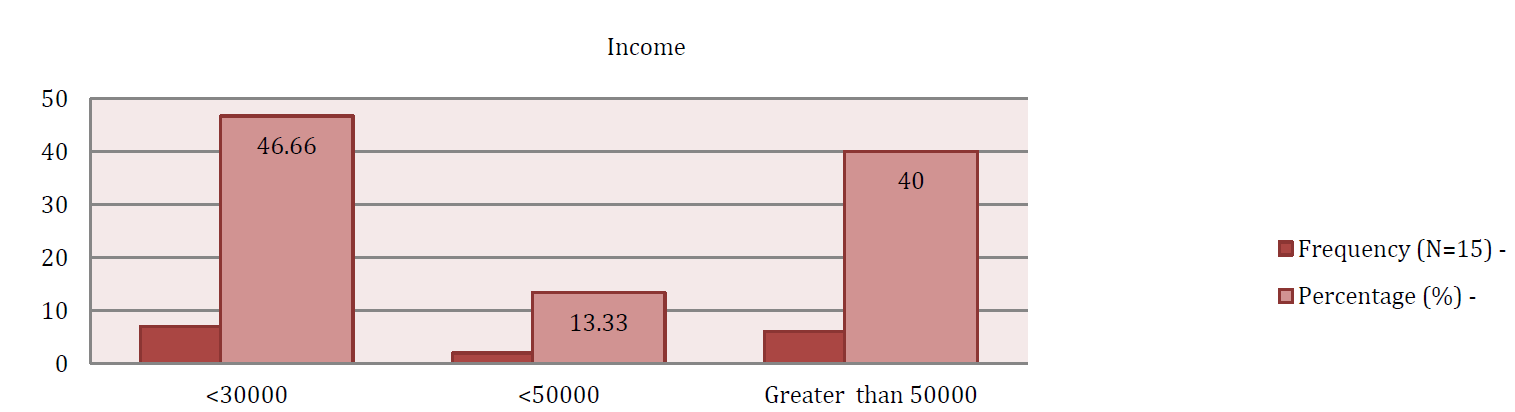
The results of the study provide valuable insights into the demographic characteristics of the participants and the associations between variables. In terms of demographic data, the mean age of the participants was approximately 46.89, with a relatively equal distribution between the age groups of 25-45 and 46-65. Similarly, the gender distribution was also nearly equal, with 64.4% male and 35.6% female participants. The mean BMI was approximately 33.24, indicating that the majority of patients (44.8%) were classified as obese.
Additionally, the study examined the frequencies of valsartan usage among the participants. Out of the total 87 participants, 63.2% received valsartan, while 36.8% did not.
The association between interleukin 6 (IL6) and other variables was assessed using Pearson correlation analysis. The results showed no significant correlations between IL6 and age or BMI. Regarding the association between valsartan and IL6, the chi-square test indicated a significant relationship with a p-value of 0.011. This suggests that there is an association between the use of valsartan and IL6 levels. However, no significant associations were found between valsartan and gender or age group. When comparing these findings with other studies, it is crucial to consider the specific research question, methodology, and population characteristics of each study. This allows for a comprehensive evaluation of the consistency or divergence of results across different studies.
The result indicating a significant association between BMI and valsartan (p = 0.001). There are several possible explanations for this significant association. First, it is plausible that the use of valsartan may lead to changes in body weight or composition, potentially resulting in alterations in BMI. Valsartan's mechanism of action in blocking angiotensin II receptors may have an influence on fluid balance, sodium excretion, or metabolic processes, which can indirectly affect BMI. Secondly, it is also possible that individuals with higher BMIs are more likely to be prescribed valsartan due to their higher risk of hypertension or cardiovascular conditions. In this case, the significant association could be reflective of the underlying health conditions associated with higher BMI rather than a direct effect of valsartan on BMI.
However, it is important to note that this study's findings are specific to the study population and should not be generalized without further research. Additionally, the nature of the association between BMI and valsartan requires more investigation to determine causality and to establish the clinical significance of this relationship.[10]
In summary, the study revealed important demographic data and associations between variables in the study population. The participants had a relatively equal distribution of age and gender. The majority of patients were classified as obese based on their BMI. The use of valsartan was associated with IL6 levels. However, to draw more robust conclusions, further research and comparisons with other studies are needed.