One of the most exciting new technologies of the twenty-first century is nanotechnology [1]. It is the capacity to apply the theory of nanoscience to practical situations by monitoring, quantifying, assembling, regulating, and producing matter at the nanoscale scale [2]. Materials with at least one dimension less than 100 nm are categorized as nanoparticles (NPs) [3]. NPs are not novel to the environment; they can be found there naturally as minerals, clays, and bacterial byproducts. Engineered nanoparticles are made to possess characteristics that are absent from large samples of the same materials. Engineered nanoparticles differ from naturally occurring ones in that they are made of a range of materials, come in a variety of sizes and forms, and have a suite of synthetic surface molecules [4]. Advances in transdisciplinary research and nanotechnology have made it possible to produce materials at the nanoscale with distinct chemical and physical characteristics, which qualify them for use in biomedical applications [5]. In general, nanotechnology refers to the synthesis and manipulation of matter at a few hundred nanometer size, which allows for the creation of certain size-dependent features [6]. For use in nanomedical applications, nanoparticles (NPs) should ideally be less than 200 nm [7]. NPs have improved colloidal stability and, thus, greater bioavailability because of their tiny size and vast surface area [8]. This indicates that NPs can pass past endothelial cells, enter the pulmonary system, and cross the blood-brain barrier [9]. In particular, metal oxide nanoparticles, or MONPs, have a number of benefits that make them a promising tool for biomedical applications [10]. These benefits include high stability, easy preparation, easy engineering to the desired size, shape, and porosity, no variations in swelling, ease of incorporation into hydrophobic and hydrophilic systems, and ease of functionalization by various molecules due to the surface's negative charge [11,12]. It is vital to define MONPs' morphology since their interactions with in vivo systems vary according on their size, shape, purity, stability, and surface characteristics [13]. MNPs may be categorized as zero-, one-, two-, or three-dimensional based on the number of dimensions, which are not limited to the nanoscale [14]. Nanoparticles, nanoclusters, quantum dots, and other materials with all of their dimensions on the nanoscale scale are examples of zero-dimensional nanomaterials. One dimension is beyond the nanoscale for one-dimensional nanoparticles (NPs), such as nanorods, nanotubes, nanowires, and nanofibers [15,16]. Generally, MONPs have highly ionic nature and can be organized with crystal morphologies exhibiting various reactive sites and corners. The deposition of MONPs on the desired position with a nanoscale resolution on a certain substrate has a promising potential to realize nanodevices for various applications in chemistry, electronics, optics and biomedicine [17]. The biomedical application of NPs for diagnostics and therapeutics (drug delivery and enhanced performance of medical devices) rapidly progresses during recent years. The usage of MONPs for diagnostics and therapy oers many advantages of modern medicine. The engineering of water-dispersible NPs allows these particles to be used in countless basic or applied biomedical researches. Right now, NPs are used in diagnostic for imaging of plentiful molecular markers, of genetic and autoimmune diseases, malignant tumors, photosensitizers in photodynamic therapy and target delivery of drugs [18]. The present study is focused on the synthesis of nickel oxide nanoparticles and study their characterization and bioactivity.
Experimental Part
Nickle (II) chloride (99%), Sodium hydroxide (NaOH) (98%), Potassium hydroxide (KOH) (98%) and ethanol (98%) were analytical grade and purchased from BDH Company (England).
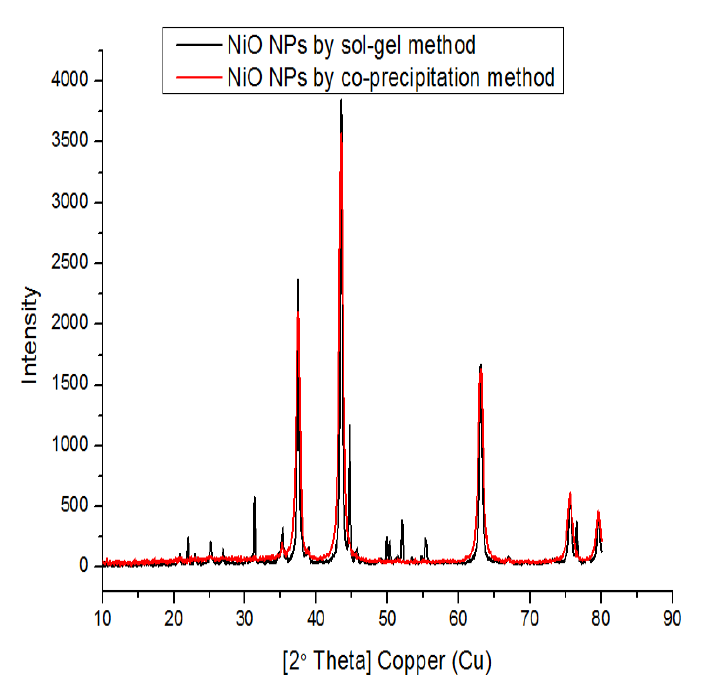
Synthesis of NiO Nanoparticles using the Co-Precipitation Method
Nickel (II) chloride (NiCl2) was used in the co-precipitation process to create NiO nanoparticles. Precursor salt (2 g) was dissolved in 75 mL deionized water, and then a 1M NaOH solution was gradually added to the salt solution while being vigorously stirred until the pH reached 12. According to the following equation, sodium hydroxide solution and nickel chloride solution reacted to form a precipitate of nickel hydroxide.
NiCl2 + 2NaOH → 2NaCl + Ni (OH)2
A green precipitate Ni(OH)2 was obtained, and it was repeatedly washed with deionized water and absolute ethanol until pH reached 7. It was then dried at 70°C for an hour. Ultimately, Ni(OH)2 is calcined for five hours at 500°C in a furnace to produce nickel oxide nanoparticles (NiO NPs). According to the following equation
Ni(OH)2 → NiO + H2O
Synthesis of NiO Nanoparticles using the Sol Gel Method
Analytical-grade reagents, including NiCl2, citric acid, C6H8O7⋅H2O (CA), n-propanol, C3H7OH, and KOH, were employed in the tests. First, a clear chelating solution was made by dissolving 0.135 mol of citric acid in 100 mL of n-propanol and stirring at 60 °C for 45 minutes. After that, this solution was agitated for 45 minutes at 60 °C, and 0.05 mol of NiCl2 was introduced. The mixture had changed into a translucent, greenish condition. After extracting the excess of the green-brown precursor propanol, the resultant solution was vacuum-heated at 90 °C to get the yield. Afterward, the sample was set in a furnace for 4 hours with a heating rate of 3.3 °C per minute at 500 °C to produce black NiO nanoparticles [19]. The products of NiO NPs were characterized using various techniques.