Thalassemia is the most common form of inherited anemia worldwide which is characterized by the decreased or abolished production of either the alpha-like (alpha-thalassemia) or the beta-like (beta- thalassemia) globin chains that are produced to form hemoglobin tetramers (alpha2gamma2, HbF: alpha2- beta2, HbA; alpha2delta2, HbA2) during the fetal and postnatal life [1].
In 1925, thalassemia was first recognized in its severe (named Cooley’s anemia) and milder (named La Malattia di Rietti-Greppi-Micheli) forms inde- pendently in the US and Italy, which are today known as thalassemia major (TM) and thalassemia.
Intermedia (TI) [2-3]. Over the next 20 years, it became clear that thalassemia had been described for homo- zygous or compound heterozygous states for a recessively inherited microcytic mild anemia terms as thalassemia minor [4]. Subsequently, it has been recognized that thalassemia resulting from defects in the production of alpha- and beta-globin chains of hemoglobin, lead to the most common monogenic disease in humans not confined to the Mediterranean, but widespread throughout the Africa, Middle East, Southeast Asia, and Western Pacific region [5]. thalassemia and HbH disease every year [6]. The extensive studies strongly indicated that the carrier status for either alpha- or beta-thalassemia offers protection against the Falciparum malaria, which explains high carrier frequency in areas where malaria has been endemic [7].
Long established screening programs for detecting carriers in the population with genetic counseling and the option of prenatal diagnosis at risk pregnancies have resulted in a marked reduction in the rate of affected birth in Mediterranean countries. Systematic carrier screening has recently been established in parts of Middle East and Asia countries.6 Following identification of both disease-causing alleles, prenatal diagnosis can be performed by analysis of DNA extracted from fetal cells obtained by either amnio- centesis or chorionic villus sampling (at 15–18 weeks and 10–12 weeks of gestation respectively). Preim- plantation genetic diagnosis would also be an option for getting pregnant to an unaffected child. It is expected that the analysis of circulating fetal DNA in maternal blood will play an increasingly important role in the future practice of prenatal diagnosis [8].
Aim of Study
To identify the relationship between adult patients with beta thalassemia and its relationship to iron overload
Literature Review
History of Thalassemia: The first clinical description of thalassemia syndrome is attributed to the Detroit pediatricians Thomas B. Cooley and Pearl Lee. The actual term thalassemia was invented by George Whipple [9]. The term “thalassemia” is derived from the Greek words (Thalassa) meaning sea and (Heam) meaning blood, and refers to disorders associated with defective synthesis of α-globin or β-globin subunit of hemoglobin [10]. Thalassemia is among the most common genetic disorders worldwide. Occurring more frequently in the Mediterranean region of Southeast Asia, and West Africa, the Indian subcontinent [11]. Through the years the population migration to new countries caused the prevalence of thalassemia to the world [12].
Thalassemia is a public hematological disease in Iraq, especially in the north region of Iraq because these regions border with Turkey and Iran which are considered as highly endemic of thalassemia and the nature of their population have mixed ethnic, in addition to consanguineous marriages. In Iraq until end of 2015, β-thalassemia reported 8246 patients in 16 thalassemia centers throughout the country that prevalence was 27.4 per 100,000 of Iraqi population, it is considered high value may be due to lack of thalassemia prevention program and consanguineous marriage [13].
Geographical Distribution
The thalassemia has a high incidence in a broad area extending from the Mediterranean basin and parts of Africa, throughout the Middle East, the Indian subcontinent, Southeast Asia, and Melanesia into the Pacific Islands [14].
The carrier frequencies for β-thalassemia in these areas range from 1 to 20%, and rarely may be higher. The frequencies for the milder forms of β-thalassemia are much greater, varying from 10 to 20% in parts of sub- Saharan Africa, to 40% or more in some Middle Eastern and Indian populations. Globally, it is estimated that there are 270 million carriers with abnormal hemoglobin and thalassemia, of which 80 million are carriers of β-thalassemia. Recent surveys suggest that between 300,000 and 400,000 babies are born with a serious hemoglobin disorder each year, 23,000 with β-thalassemia major [15].
Types of Thalassemia
Thalassemia is an inherited disorder caused by impaired synthesis of one or more globin chains. This type of hereditary anemia is caused by decreased or absent production of one type of globin chains either α or β globin chain. They are divided according to which globin chains are produced in reduced amounts into the:
All types of thalassemia are considered quantitative hemoglobin disease. Only the α and β thalassemia are sufficiently common to be of importance[11,15].
Alpha-Thalassemia
Alpha-thalassemia is usually produced by one or more deletions of the alpha globin chains. It is generally presented as a milder form of the disease. This is due to the fact that there are four α-globin genes requiring multiple mutations to result in a clinical impact [16]. The clinical syndromes resulting from α-thalassemia are first recognized in the mid-1950 and early 1960 as hypochromic microcytic anemia in the absence of iron deficiency [17-18].
Beta Thalassemia
Beta thalassemia is the result of a defective or absent synthesis of beta globin chains of the globin [19]. It is the most important type amongst the thalassemia syndrome and has become a worldwide clinical problem due to an increasing immigrant population [20].
Hypochromia and microcytosis characterize all forms of beta-thalassemia. The beta-thalassemia can be divided into several varieties. In β° thalassemia there is a total absence of β-chain production. In β+ thalassemia, there is a partial deficiency of beta-chain production [18].
The clinical severity of beta- thalassemia is related to the extent of imbalance between the alpha-globin and beta-globin chains [21].
Classification of Beta Thalassemia
Clinically β-thalassemia can be classified into three major groups:
Beta Thalassemia Major
Thalassemia major, also known as Cooley’s anemia and the Mediterranean anemia is the most severe form of β-thalassemia since both mutations of both β-globin alleles result in severely impaired β-globin chain production [22].
Three of the general allele combinations are responsible for thalassemia phenotype - βo/βo, βo/β+, and sometimes β+/β+ [23]. In thalassemia major the excess unpaired alpha- globin chain aggregate to form inclusion bodies.
These inclusion bodies damage RBC membranes, leading to intravascular hemolysis [24].
In addition, there is damage and premature destruction of RBC precursors, causing ineffective erythropoiesis. Anemia is severe and oxygen transport is compromised [25].
In some patients, death would result without chronic blood transfusions. Other clinical manifestations include listlessness, fatigue, dyspnea, poor appetite, hepatosplenomegaly, heart failure, and bone deformation and delayed puberty [25].
Beta Thalassemia Intermedia
Patients with β-thalassemia intermedia have mild to moderate anemia and in most cases do not require blood transfusions [26].
This condition is milder than thalassemia major due to an inheritance of a β- globin gene mutation associated with reduced β-globin chain production. The reduced genotype is most commonly β+/β+ [17].
The clinical phenotype of thalassemia intermedia is approximately intermediate between thalassemia major and minor. The common clinical features and complication include spleen enlargement due to entrapment of damaged RBCs, with a risk of iron overload due in part to increased intestinal absorption [27].
Although thalassemia intermedia can be associated with poor growth and bone abnormalities, it presents later in life and rarely affects longevity [26].
Patients require regular monitoring because the clinical severity varies widely between patients and within a patient over time, with possible deterioration to the thalassemia major phenotype [28]. Several rare β-globin variants phenotypically manifest thalassemia intermedia. For instance, individuals with dominantly inherited β-thalassemia or inclusion body β-thalassemia clinically exhibit thalassemia intermedia. These patients have moderate anemia and splenomegaly [29]. A rare variant form called “silent β-thalassemia” results from a mild imbalance of globin chain synthesis due to reduced β-globin synthesis, leading to thalassemia intermedia. Silent β-thalassemia mutations are found mainly in the regulatory regions, β-globinpromoter and 5’ and 3’ UTRs[26].
Beta Thalassemia Minor
Thalassemia minor is the most common form of β- thalassemia, and also known as the ‘thalassemia trait, in which affected individuals are asymptomatic [30].
These forms are typically heterozygous for β-thalassemia since they carry one normal hemoglobin β- globin allele and one thalassemia allele - either βoor β+ [31].
The asymptomatic patients are usually detected through routine hematological tests, but in retrospect, some newly diagnosed patients are observed to have slight anemia and small RBCs. The primary warning for individuals with thalassemia minor is a potential uncertainty of having children changed with more serious thalassemia if their wife or vice versa is also a carrier of thalassemia minor [32].
Diagnosis of Thalassemia
The initial investigations in a patient with suspected thalassemia at presentation consist of a complete blood count (CBC) or blood picture. Characteristic features in an CBC are low hemoglobin level, mean cell volume (MCV), mean cell hemoglobin (MCH), mean cell hemoglobin concentration (MCHC), and hematocrit (HCT) levels with increased redcell distribution width (RDW). The blood picture will reveal variations in the shape and size of red cells (anisocytosis and poikilocytosis) with predominantly hypochromic microcytic cells, and nucleated red blood cells [33].
Presence of hypochromic, often wrinkled and folded cells (lepto-cells) containing irregular inclusion bodies of precipitated α globin chains is a characteristic feature in the blood film. These inclusions are prominent in splenectomised patients. Basophilic stippling, Pappeheimer bodies, and target cells are also present. The low reticulocyte count present in these patients indicates the severity of intramedullary erythroblast destruction [34].
Leukocytosis with increased neutrophil count is usually noted. In patients with hyper-spleenism due to massive splenomegaly, leucopenia with thrombocytopenia can also be seen. Bone marrow examination, although not routinely performed, will show gross erythroid hyperplasia with the reversed myeloid/erythroid ratio from the normal 3 or 4 to 0.1 or less [33].
Dis-erythropoiesis with nuclear lobulation and fragmentation, basophilic stippling, defective hemoglobinization, and α chain precipitates are also visible in the bone marrow aspirate [35].
As a result of precipitated α chains in erythroid precursors, accelerated apoptosis is noted in the poly-chromatophilic and orthochromatic stages[21]. Bone marrow iron stores are increased and active phagocytic macrophages and pseudo Gaucher cells are also present. Diagnosis of β thalassemia major is depended on the finding of the relative levels of mainly adult hemoglobin (HbA) and fetal hemoglobin (HbF) using hemoglobin electrophoresis, isoelectric focusing or High-Performance Liquid Chromatography (HPLC) methods. The findings will be varied according to the functional hemoglobin allele which is present in the patient; β° and β+ alleles. In homozygous or compound heterozygous for β° allele (β°β°), HbA is absent, the predominant type is HbF (approx.95-98%) and HbA2 can be variable. When there is a homozygous state for β+ allele (β+β+) or compound heterozygous state for β° and β+ alleles (β°β+) HbA is also present in variable amounts; can be up to 35% of total hemoglobin [11,33]. High serum ferritin and saturated iron binding capacity are signs of iron overload and seen in most patients with transfusion-dependent thalassemia. This change is reflected as increased iron in both reticuloendothelial and parenchymal cells in the liver and increased bone marrow iron stores.
Bilirubin level is usually elevated and haptoglobins are absent. Serum aspartate aminotransferase (AST) levels are frequently high at the time of diagnosis due to hemolysis. Alanine aminotransferase (ALT) levels are usually normal prior to transfusion therapy and subsequently, ALT levels may elevate due to iron-induced hepatic damage or viral hepatitis [36].
Assessment of Iron Overload and its Effect on Liver Enzymes
Ferritin is the iron storage protein that serves to store iron in a non-toxic form, to store it in a safe form, and to transport it to any site in the body where it is required. Ferritin level in serum directly relates to the amount of iron stored in the body, which is important for red blood cell production [37]. If ferritin is in height level, there is iron in excess. Ferritin levels are also used as specific indicators for iron overload. Normal ranges of ferritin are 12–300 and 12–150 ng/ ml for male and female, respectively [38]. A significantly high level of ferritin is found in patients with iron overload as this may help differentiate thalassemia patients from those with iron deficiency, both of which will have a low red blood cell count [39]. In addition, any inflammatory state can cause a high level of ferritin; it acts as an acute phase protein. Therefore, long term monitoring of ferritin would be necessary, to gain any additional information for thalassemia diagnosis or treatment of follow-up patients [22].
Alanine Transaminase (ALT), and Aspartate. Transaminase
Alanine transaminase ALT, and aspartate transaminase AST are enzymes placed hepatic cell that released into the bloodstream when liver cells are damaged, these two enzymes are previously known as the GPT serum glutamic-pyruvic transaminase and the GOT serum glutamate- oxaloacetate transaminase [40].
These two transaminase enzymes may be reported on laboratory with both their new names and previous names or by their newer names only, ALT and AST are present in highest concentrations in cells from the heart, liver, skeletal muscles and red blood cells. Patients whose liver function test show a predominant rise in the transaminases have liver diseases that are characterized by hepatocellular damage [41].
ALT is found predominantly in the liver, with lesser quantities found in the kidneys, heart and skeletal muscle, as a result the ALT is a more specific-indicator of liver inflammation than the AST, as the AST may also be elevated in diseases affecting other organs such as the heart or muscles as in the following equation.
Alanine + α-ketoglutarate ⇌ pyruvate + glutamate
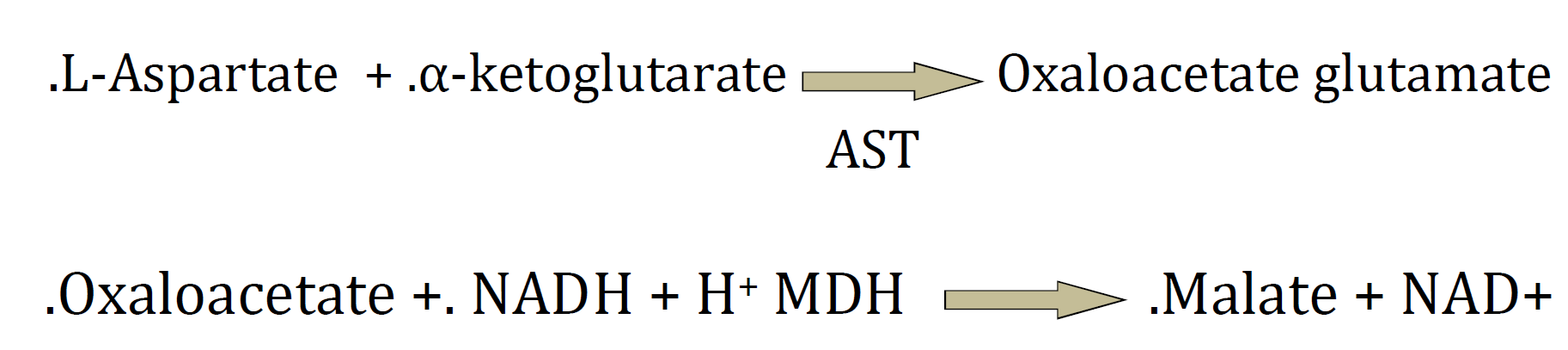
The AST is also elevated after a myocardial infarction, and during acute pancreatitis, acute hemolytic anemia, severe burns, acute renal disease, musculoskeletal diseases, and trauma. In contrast, aspartate transaminase is responsible for catalyzes the interconversion of aspartate and α- ketoglutarate to oxaloacetate and glutamate [42].
Aspartate + α-ketoglutarate ↔ oxaloacetate + glutamate
Levels of aminotransferases (ALT) in the blood indicate the degree to which liver membrane damage has resulted in an increased release of hepatocellular enzyme into the bloodstream, because ALT is more specific than AST for liver damage, ALT is used more often, in patients with risk factors for HCV infection and in whom there is no another explanation for increased enzyme levels, elevated aminotransferase levels are highly associated with HCV infection [33,43].
Alkaline Phosphatase (ALP)
ALP is present in mucosal epithelia of the small intestine, proximal convoluted tubules of the kidney, placenta, liver and bone. It performs lipid transportation in the intestine and calcification in bone. The activity of serum ALP is mainly from the liver with 50% contributed by bone, normal serum ALP is 41 to 133U/ml. In acute viral hepatitis, ALP usually remains normal or moderately increases. Elevation of ALP with prolonged itching is related with viral hepatitis A presenting cholestasis, tumor cells secrete ALP into plasma and there are tumor specific isoenzymes [44].
Hepatic and bony metastasis can also cause elevated levels of ALP, other diseases like infiltrative liver diseases, abscesses, granulomatous liver disease and amyloidosis may cause a rise in ALP, mildly elevated levels of ALP may be seen in liver cirrhosis, hepatitis and congestive cardiac failure [45].
Low levels of ALP occur in hypothyroidism, pernicious anemia, zinc deficiency and congenital hypo- phosphatasia, ALP has been found elevated in peripheral arterial disease, independent of other traditional cardiovascular risk factors [45].
Thalassemia Program in Iraq
This program was established in 1989, under the responsibility of the Ministry of health and in collaboration with WHO. It provides all the medical services for thalassemia management and control programs. There are more than 7000 thalassemia patients registered in 6 thalassemia center’s they are Baghdad, Ninawa, Waist, Bassrah, and Babylon and 8 clinics unit including Salah Din, Diyala, Messan, Thi-Qar, Muthana, Al-Najaf, Karbala, and Al- Anbar. All their medical services are free of charge. These centers are responsible to provide:
Adequate and safe blood supply for the patients in collaboration with the central blood bank
Adequate medical services for the management of thalassemia center including doctors, nurses, health-workers and laboratory workers
Training for medical staff by training courses, workshops, seminars, conferences, and encouraging research
Prevention programs, which include:
Family planning by providing the patients with contraceptive materials
Educational programs for the health workers, families, and the general population to be aware of how to control disease by interviews, folders, posters, TV spots etc
Carrier identification: in 1999 detection of the carrier rate among secondary school students was 4.8%
There are two projects which have been decided by the scientific committee to be done:
Premarital screening and introduction of a premarital test of thalassemia in the premarital certificate
Prenatal diagnosis, there is a trained medical team for this project is available but not feasible now because of the lack of kits for molecular studies
Evaluation by maintaining registration of patients, and report progress of patient care from time to time