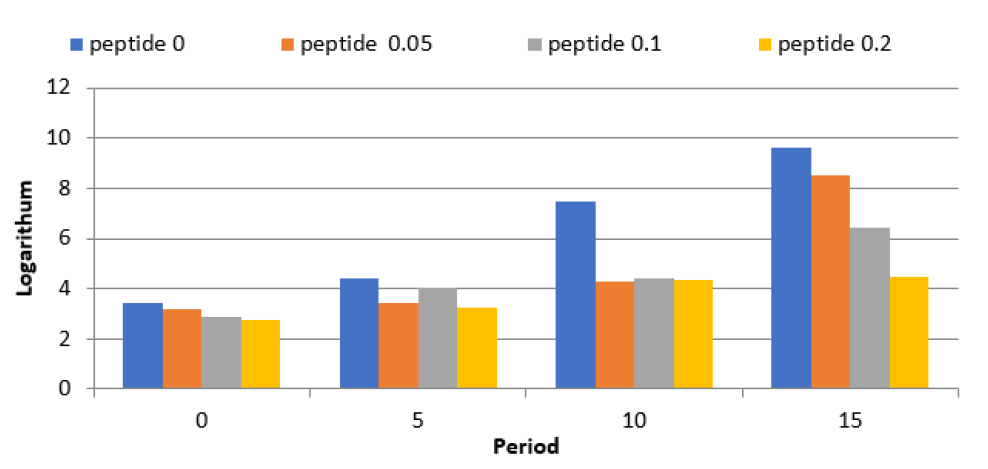
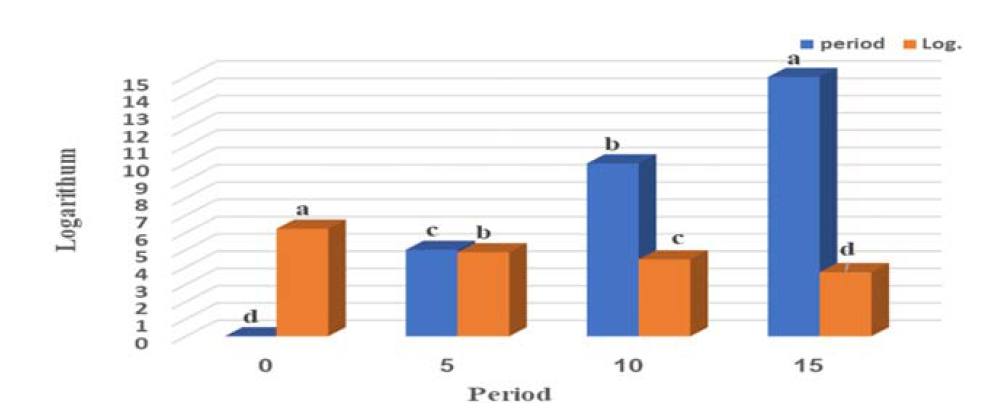
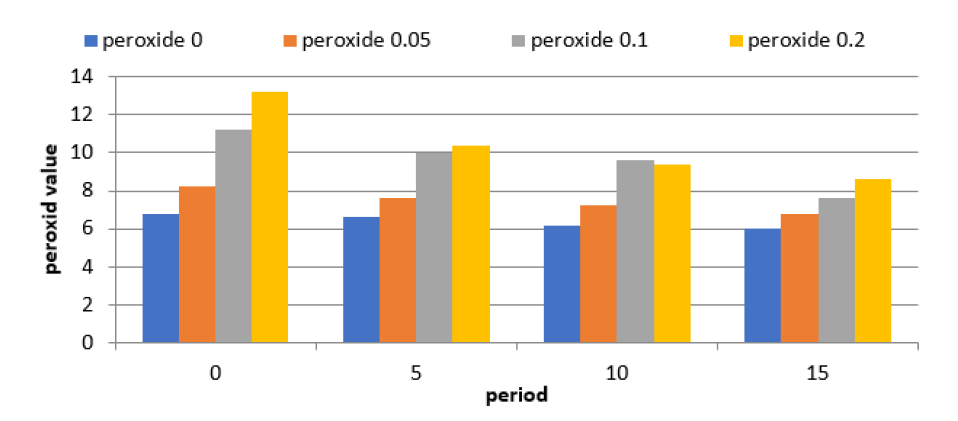
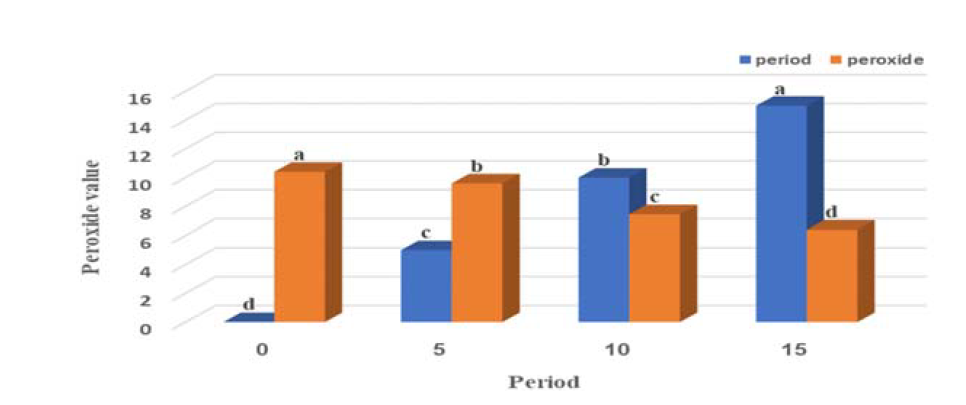
Barnes, L., editor. Surgical Pathology of the Head and Neck. 3rd ed., vol. 1.
Damjanov, I., and J. Linder. Anderson’s Pathology. vol. 2.
Ellis, G.L. et al. Surgical Pathology of the Salivary Glands. Major Problems in Pathology, vol. 25.
“Minor Salivary Gland Mucoepidermoid Carcinoma in Children and Adolescents: A Case Series and Review of the Literature.” Journal of Medical Case Reports, vol. 6, 2012, p. 182. https://doi.org/10.1186/1752-1947-6-182.
Gupta, R. et al. “Salivary Gland Lesions: Recent Advances and Evolving Concepts.” Journal of Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology, vol. 119, no. 6, June 2015.
Nagao, T. et al. “Dedifferentiation in Low-Grade Mucoepidermoid Carcinoma of the Parotid Gland.” Human Pathology, vol. 34, no. 10, October 2003.
Ambika, L. “Low-Grade Mucoepidermoid Carcinoma of the Tongue: An Unusual Presentation.” Journal of the Scientific Society, vol. 39, no. 2, May–August 2012.
“Review and Updates of Immunohistochemistry in Selected Salivary Gland and Head and Neck Tumors.” Archives of Pathology and Laboratory Medicine, vol. 139, January 2015.
Weinreb, I. et al. “Oncocytic Mucoepidermoid Carcinoma: Clinicopathologic Description of 12 Cases.” American Journal of Surgical Pathology, vol. 33, no. 3, 2009, pp. 409–416. https://doi.org/10.1097/PAS.0b013e318184b36d.
Sam’s, R.N., and D.R. Gnepp. “p63 Expression Can Be Used in Differential Diagnosis of Salivary Gland Acinic Cell and Mucoepidermoid Carcinomas.” Head and Neck Pathology, vol. 7, no. 1, 2013, pp. 64–68.
“Mucoepidermoid Carcinoma of Salivary Glands in the Pediatric Age Group: 18 Cases, Including 11 Second Malignant Neoplasms.” Head & Neck, 2006. https://doi.org/10.1002/hed.20429.
Dorso, L. et al. “High-Grade Mucoepidermoid Carcinoma of the Mandibular Salivary Gland in a Lion.” Veterinary Pathology, vol. 45, 2008, pp. 104–108.
Kolude, B. et al. “Mucoepidermoid Carcinoma of the Oral Cavity.” Journal of the National Medical Association, vol. 93, no. 5, May 2001.
Nakayama, T. et al. “Clinicopathological Significance of the CRTC3-MAML2 Fusion Transcript in Mucoepidermoid Carcinoma.” Modern Pathology, vol. 22, 2009, pp. 1575–1581. https://doi.org/10.1038/modpathol.2009.113.
Fonseca, F.P. et al. “p63 Expression in Papillary Cystadenoma and Mucoepidermoid Carcinoma of Minor Salivary Glands.” Journal of Oral and Maxillofacial Pathology, vol. 115, no. 1, January 2013.
Martínez-Ollero, J. et al. “Nasopharyngeal Mucoepidermoid Carcinoma: A Case Report and Review of Literature.” Reports of Practical Oncology and Radiotherapy, vol. 18, 2013, pp. 117–120.
Simon, D. et al. “Central Mucoepidermoid Carcinoma of Mandible: A Case Report and Review of the Literature.” World Journal of Surgical Oncology, 2003, pp. 1–5.
Munde, A. et al. “Central Mucoepidermoid Carcinoma of the Mandible.” Indian Journal of Dental Research, vol. 21, no. 4, 2010.
Sepúlveda, I. et al. “Mandibular Central Mucoepidermoid Carcinoma: A Case Report and Review of the Literature.” Case Reports in Oncology, vol. 7, 2014, pp. 732–738.
Tucci, R. et al. “Central Mucoepidermoid Carcinoma: Report of a Case with 11 Years’ Evolution.” Medicina Oral, Patología Oral y Cirugía Bucal, vol. 14, no. 6, June 2009, pp. E283–E286.
Kanmani, R., and M.J. Daniel. “Intraosseous Mucoepidermoid Carcinoma of the Maxilla: A Silent Invader.” Indian Journal of Oral Sciences, vol. 5, no. 1, January–April 2014, pp. 39–43.
Mahesh, K. et al. “Cytological Diagnosis of Mucoepidermoid Carcinoma of Parotid – A Diagnostic Dilemma.” International Journal of Medical Science and Public Health, vol. 2, no. 2, 2013, pp. 462–464.
Jindal, T. et al. “Role of PET-CT in Bronchial Mucoepidermoid Carcinomas: A Case Series and Review of the Literature.” Journal of Medical Case Reports, vol. 4, 2010, p. 277. https://doi.org/10.1186/1752-1947-4-277.
Kim, Y.S. et al. “Immunohistochemical Array for Clear Cell Type Mucoepidermoid Carcinoma.” The Korean Journal of Pathology, vol. 44, 2010, pp. 284–294.
Bharathi, U. et al. “Mucoepidermoid Carcinoma of Palate: A Case Report.” IJSS Case Reports & Reviews, vol. 1, no. 7, December 2014.
Seyedmajidi, M., and R. Foroughi. “Central Mucoepidermoid Carcinoma: A Case Report and Review of the Literature.” Journal of Clinical & Experimental Oncology, vol. 2, no. 2, 2013.
Kim, Y.S. et al. “Immunohistochemical Array for Clear Cell Type Mucoepidermoid Carcinoma.” The Korean Journal of Pathology, vol. 44, 2010, pp. 284–294.
(Duplicate of #24 — same reference)
Gautam, N.S. et al. “Mucoepidermoid Carcinoma of Buccal Mucosa.” BMJ Case Reports, 2014.
Lawlor, C.M. et al. “A Rare Case of Mucoepidermoid Carcinoma of the Lingual Tonsil with Cervical Lymph Node Metastases.” Journal of Cancer Therapeutics & Research, vol. 4, 2015.