Lahariya, C., Goel, M. K., Kumar, A., Puri, M., & Sodhi, A. “Emergence of viral hemorrhagic fevers: Is recent outbreak of crimean congo hemorrhagic fever in India an indication?”. Journal of Postgraduate Medicine (2012): 39 https://www.researchgate.net/publication/221885941
Watts, D. M., Ksiazek, T. G., Linthicum, K. J., & Hoogstraal, H. “Crimean-Congo hemorrhagic fever”. The Arboviruses (2019): 177-222
Ghareeb, O. A., & Ali, Q. A. “Waterborne Zoonotic Bacterial Pathogens”. Texas Journal of Medical Science (2023): 63-69 https://doi.org/10.62480/tjms.2023.vol21.pp63-69
Ghareeb, O. A., & Sultan, A. I.” Nipah-An Emerging Viral Zoonotic Disease: A Review”. Annals of the Romanian Society for Cell Biology (2021): 456-465 http://annalsofrscb.ro
Ghareeb, O. A. “ Ebola-A fatal Emerging Zoonotic Disease: A Review”. Annals of the Romanian Society for Cell Biology (2021): 8748-8754 http://annalsofrscb.ro
Kzar, A. J., Faiq, T. N., & Ghareeb, O. A. “Recent infection with black fungus associated with COVID-19: a review”. Pakistan Journal of Medical and Health Sciences (2021): 1771-1773 https://doi.org/10.53350/pjmhs211551771
Al-Haidari, K. A., Faiq, T. N., & Ghareeb, O. A. “Preventive value of black seed in people at risk of infection with COVID–19”. Pakistan J Med Health Sci (2021): 384-387 https://pjmhsonline.com
Ciloglu, A., Ibis, O., Yildirim, A., Aktas, M., Duzlu, O., Onder, Z., & Inci, A. “Complete mitochondrial genome characterization and phylogenetic analyses of the main vector of Crimean-Congo haemorrhagic fever virus: Hyalomma marginatum Koch, 1844”.Ticks and Tick-Borne Diseases (2021): 101736 https://www.sciencedirect.com
Alhilfi, R. A., Khaleel, H. A., Raheem, B. M., Mahdi, S. G., Tabche, C., & Rawaf, S.”Large outbreak of Crimean-Congo haemorrhagic fever in Iraq, 2022”. IJID regions (2023): 76-79 https://www.ncbi.nlm.nih.gov
Rodriguez, S. E., Hawman, D. W., Sorvillo, T. E., O'Neal, T. J., Bird, B. H., Rodriguez, L. L.& Spengler, J. R.” Immunobiology of Crimean-Congo hemorrhagic fever”. Antiviral research (2022): 105244 https://pubmed.ncbi.nlm.nih.gov
Messina, J. P., Pigott, D. M., Golding, N., Duda, K. A., Brownstein, J. S., Weiss, D. J. & Hay, S. I. “The global distribution of Crimean-Congo hemorrhagic fever”. Transactions of the Royal Society of Tropical Medicine and Hygiene (2015) 503-513 https://doi.org/10.1093/trstmh/trv050
Sah, R., Mohanty, A., Mehta, V., Chakraborty, S., Chakraborty, C., & Dhama, K. “Crimean-Congo haemorrhagic fever (CCHF) outbreak in Iraq: Currently emerging situation and mitigation strategies–Correspondence”. International Journal of Surgery (2022): 106916 https:// DOI: 10.1016/j.ijsu.2022.106916
Spengler, J. R., Bergeron, É., & Rollin, P. E.” Seroepidemiological studies of Crimean-Congo hemorrhagic fever virus in domestic and wild animals”. PLoS neglected tropical diseases (2016): e0004210 https://doi.org/10.1371/journal.pntd.0004210
Schmaljohn, C. S., and Hooper, J. W. “in Bunyaviridae: the Viruses and Their Replication, 4th Ed. Fields Virology (Knipe, D. M., and Howley,P. M. eds.), Lippincott, Williams & Wilkins, Philadelphia”(2001): https://www.sciencedirect.com › topics › neuroscience
Sanchez, A. J., Vincent, M. J., Erickson, B. R., & Nichol, S. T. “Crimean-congo hemorrhagic fever virus glycoprotein precursor is cleaved by Furin-like and SKI-1 proteases to generate a novel 38-kilodalton glycoprotein”. Journal of virology (2006): 514-525 https://doi.org/10.1128/jvi.80.1.514-525.2006
Altamura, L. A., Bertolotti-Ciarlet, A., Teigler, J., Paragas, J., Schmaljohn, C. S., & Doms, R. W. “Identification of a novel C-terminal cleavage of Crimean-Congo hemorrhagic fever virus PreGN that leads to generation of an NSM protein”. Journal of virology (2007): 6632-6642 https://doi.org/10.1128/jvi.02730-06
Sánchez-Seco, M. P., Sierra, M. J., Estrada-Peña, A., Valcárcel, F., Molina, R., de Arellano, E. R., & Negredo, A. “Widespread detection of multiple strains of Crimean-Congo hemorrhagic fever virus in ticks, Spain”. Emerging Infectious Diseases (2022) 394 http://doi.10.3201/eid2802.211308
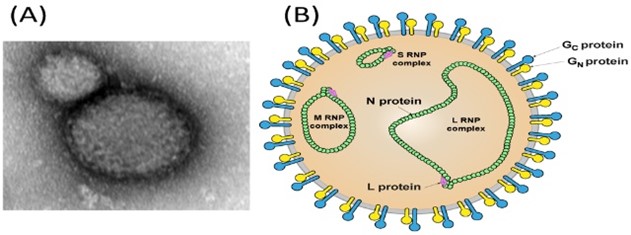
Wang, W., Liu, X., Wang, X., Dong, H., Ma, C., Wang, J., ... & Guo, Y. “Structural and functional diversity of nairovirus-encoded nucleoproteins”. Journal of virology (2015): 11740-11749 https://doi.org/10.1128/jvi.01680-15
Bente, D. A., Forrester, N. L., Watts, D. M., McAuley, A. J., Whitehouse, C. A., & Bray, M. “Crimean-Congo hemorrhagic fever: history, epidemiology, pathogenesis, clinical syndrome and genetic diversity”. Antiviral research (2013): 159-189 https://pubmed.ncbi.nlm.nih.gov
Olaya, C., Adhikari, B., Raikhy, G., Cheng, J., & Pappu, H. R. “Identification and localization of Tospovirus genus-wide conserved residues in 3D models of the nucleocapsid and the silencing suppressor proteins”. Virology journal (2019) 1-15 7 https://doi.org/10.1186/s12985-018-1106-4
Perveen, N., & Khan, G. “Crimean–Congo hemorrhagic fever in the Arab world: a systematic review”. Frontiers in Veterinary Science (2022): 938601 https://doi.org/10.3389/fvets.2022.938601
Spengler, J. R., Bergeron, É., & Rollin, P. E. “Seroepidemiological studies of Crimean-Congo hemorrhagic fever virus in domestic and wild animals”. PLoS neglected tropical diseases (2016): e0004210 https://doi.org/10.1371/journal.pntd.0004210
Papa, A., Tsergouli, K., Tsioka, K., & Mirazimi, A. “Crimean-Congo hemorrhagic fever: tick-host-virus interactions”. Frontiers in cellular and infection microbiology (2017): 213 https://doi.org/10.3389/fcimb.2017.00213
Aradaib, I. E., Erickson, B. R., Mustafa, M. E., Khristova, M. L., Saeed, N. S., Elageb, R. M., & Nichol, S. T. “Nosocomial outbreak of Crimean-Congo hemorrhagic fever, Sudan”. Emerging infectious diseases (2010): 837 http://doi. 10.3201/eid1605.091815
Sorvillo, T. E., Rodriguez, S. E., Hudson, P., Carey, M., Rodriguez, L. L., Spiropoulou, C. F., ... & Bente, D. A. “Towards a sustainable one health approach to crimean–congo hemorrhagic fever prevention: Focus areas and gaps in knowledge”. Tropical Medicine and Infectious Disease (2020): 113 https://doi.org/10.3390/tropicalmed5030113
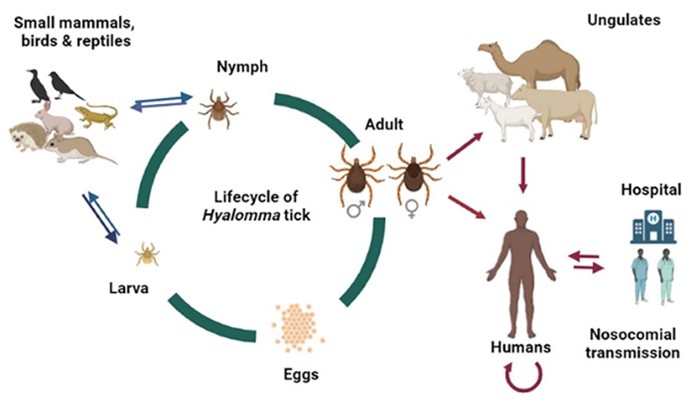
Gargili, A., Estrada-Peña, A., Spengler, J. R., Lukashev, A., Nuttall, P. A., & Bente, D. A. “The role of ticks in the maintenance and transmission of Crimean-Congo hemorrhagic fever virus: A review of published field and laboratory studies”. Antiviral research (2017): 93-119 http://doi. 10.1016/j.antiviral.2017.05.010
Shepherd, A. J., Leman, P. A., & Swanepoel, R. “Viremia and antibody response of small African and laboratory animals to Crimean-Congo hemorrhagic fever virus infection”. The American journal of tropical medicine and hygiene (1989): 541-547 https://doi.org/10.4269/ajtmh.1989.40.541
Shepherd, A. J., Swanepoel, R., Leman, P. A., & Shepherd, S. P. “Field and laboratory investigation of Crimean-Congo haemorrhagic fever virus (Nairovirus, family Bunyaviridae) infection in birds”. Transactions of the Royal Society of Tropical Medicine and Hygiene (1987): 1004-1007 http://doi.10.1016/0035-9203(87)90379-8.
Hawman DW.”Feldmann H. Crimean–Congo haemorrhagic fever virus”. Nature Reviews Microbiology (2023): 463-477
https://doi.org/10.3390/diagnostics13162708
Spengler, J. R., Estrada-Peña, A., Garrison, A. R., Schmaljohn, C., Spiropoulou, C. F., Bergeron, É., & Bente, D. A. “A chronological review of experimental infection studies of the role of wild animals and livestock in the maintenance and transmission of Crimean-Congo hemorrhagic fever virus”. Antiviral research (2016): 31-47 http://doi. 10.1016/j.antiviral.2016.09.013
Serretiello, E., Astorri, R., Chianese, A., Stelitano, D., Zannella, C., Folliero, V., ... & Galdiero, M. “The emerging tick-borne Crimean-Congo haemorrhagic fever virus: A narrative review”. Travel Medicine and Infectious Disease (2020): 10187 http://doi. 10.1016/j.tmaid.2020.101871
F. de la Calle-Prieto, A. Martín-Quir´os, E. Trigo, M. Mora-Rillo, M. Arsuaga,M. Díaz-Men´endez, J.R. Arribas.” Therapeutic management of Crimean-Congo-haemorrhagic fever”. Enfermedades infecciosas y microbiologia clinica (English ed.) (2018): 517–522 http://doi: 10.1016/j.eimce.2017.04.016
Hawman, D. W., Haddock, E., Meade-White, K., Williamson, B., Hanley, P. W., Rosenke, K., & Feldmann, H.”Favipiravir (T-705) but not ribavirin is effective against two distinct strains of Crimean-Congo hemorrhagic fever virus in mice”. Antiviral research (2018): 18-26 http://doi. 10.1016/j.antiviral.2018.06.013
Fillâtre, P., Revest, M., & Tattevin, P. “Crimean-Congo hemorrhagic fever: An update”. Medecine et maladies infectieuses (2019): 574-585 https://www.sciencedirect.com
Sharma, S. N., Singh, R., Balakrishnan, N., Kumawat, R., & Singh, S. K. “Vectors of Crimean-Congo hemorrhagic fever (CCHF): prevention and its control. Journal of Communicable Diseases (2020): 22-26 https://orcid.org/0000-0001-8569-1661
Al-Rubaye, D., Al-Rubaye, T. S., Shaker, M., & Naif, H. M. “Recent outbreaks of crimean–congo hemorrhagic fever (CCHF) In Iraq”. Sci Arch, (2022): 109-112 http://doi.org/10.47587/SA.2022.3205
Fletcher, T. E., Gulzhan, A., Ahmeti, S., Al-Abri, S. S., Asik, Z., Atilla, A., & Leblebicioglu, H. “Infection prevention and control practice for Crimean-Congo hemorrhagic fever—A multi-center cross-sectional survey in Eurasia”. PloS one (2017):e0182315 https://doi.org/10.1371/journal.pone.0182315
Mohammed, T. A., Mostafa, A. H., Ahmad, J. Q., Yahya, N. B., & Tayib, G. A. “The Trojan horse feature of SARS-CoV-2 behind the re-emergence of the Crimean-Congo hemorrhagic fever in Iraq”. Human Vaccines & Immunotherapeutics (2022): 2128610 https://doi.org/10.1080/21645515.2022.2128610
- Norouzi, M., Dayer, M. S., & Ghaffarifar, F. “Molecular detection and characterisation of Theileria in hard ticks of small ruminants in Zarrin Dasht County, Southern Iran”. Veterinary Medicine and Science (2023): 372-379 https://doi.org/10.1002/vms3.1027