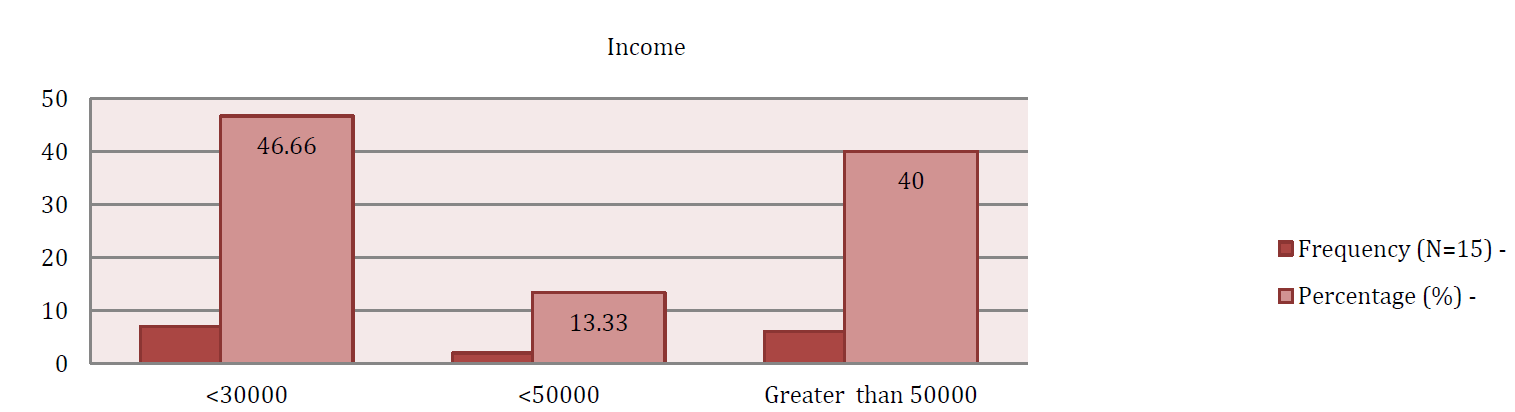
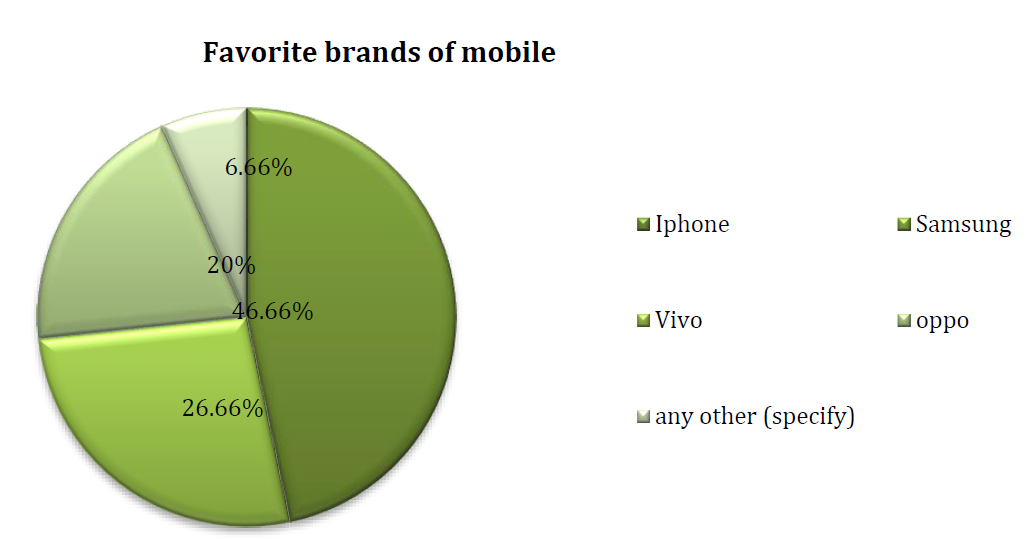
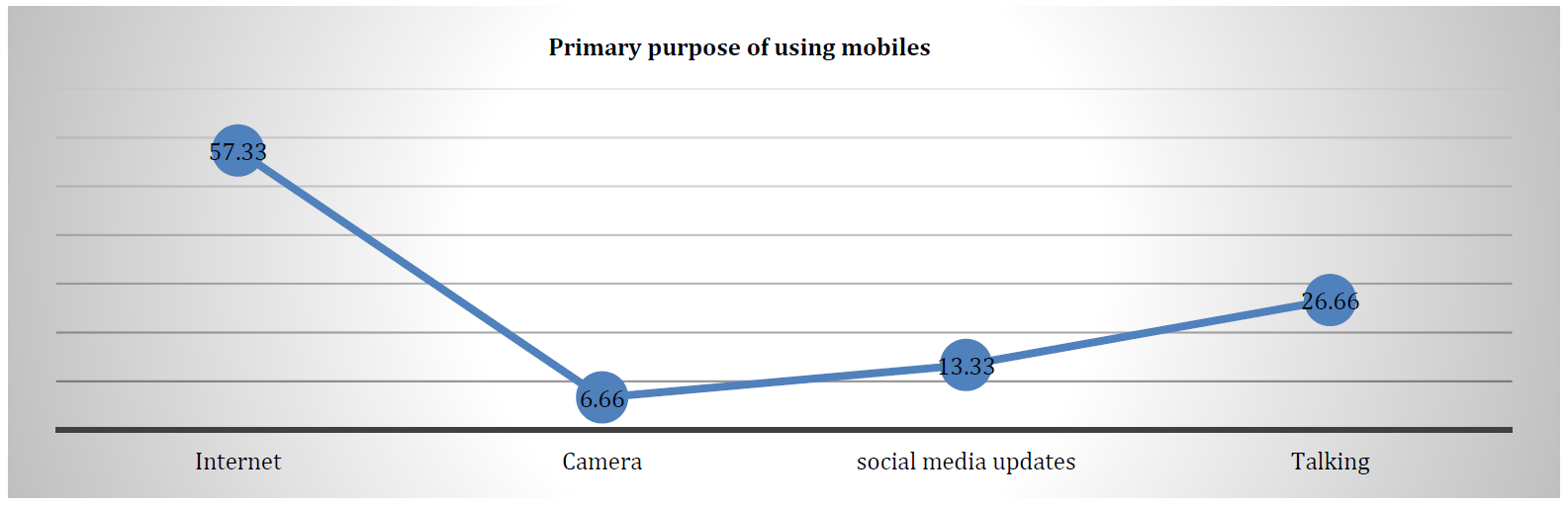
Frias J.P.G.L. and Nash R. “Microplastics: finding a consensus on the definition.” Marine Pollution Bulletin, vol. 138, 2019, pp. 145-147.
Andrady A.L. “The plastic in microplastics: a review.” Marine Pollution Bulletin, vol. 119, no. 1, 2017, pp. 12-22.
Thompson R.C. “Plastic debris in the marine environment: consequences and solutions.” Marine Nature Conservation in Europe, vol. 193, 2006, pp. 107-115.
Barnes S.J. “Understanding plastics pollution: the role of economic development and technological research.” Environmental Pollution, vol. 249, 2019, pp. 812-821.
Jambeck J. et al. “Plastic waste inputs from land into ocean.” Marine Pollution Bulletin, vol. 347, no. 6223, 2015, pp. 3-6.
Alomar C. and Deudero S. “Microplastics in the Mediterranean Sea: deposition in coastal shallow sediments, spatial variation and preferential grain size.” Marine Environmental Research, vol. 115, 2016, pp. 1-10.
Auta H.S. et al. “Distribution and importance of microplastics in the marine environment: a review of the sources, fate, effects, and potential solutions.” Environment International, vol. 102, 2017, pp. 165-176.
Pham C.K. et al. “Plastic ingestion in oceanic-stage loggerhead sea turtles (Caretta caretta) off the North Atlantic subtropical gyre.” Marine Pollution Bulletin, vol. 121, nos. 1-2, 2017, pp. 222-229.
Anastasopoulou A. et al. “Plastic debris ingested by deep-water fish of the Ionian Sea (Eastern Mediterranean).” Deep Sea Research Part I: Oceanographic Research Papers, vol. 74, 2013, pp. 11-13.
Denuncio P. et al. “Plastic ingestion in Franciscana dolphins, Pontoporia blainvillei (Gervais and d’Orbigny, 1844), from Argentina.” Marine Pollution Bulletin, vol. 62, no. 8, 2011, pp. 1836-1841.
Chen Y. et al. “Single-use plastics: production, usage, disposal, and adverse impacts.” Science of the Total Environment, vol. 752, 2020, pp. 141772-141772.
Moser M.L. and Lee D.S. “A fourteen-year survey of plastic ingestion by western North Atlantic seabirds.” Colonial Waterbirds, vol. 15, no. 1, 1992, pp. 83-94.
Cole M. et al. “Isolation of microplastics in biota-rich seawater samples and marine organisms.” Scientific Reports, vol. 4, 2011, p. 4528.
Fendall L.S. and Sewell M.A. “Contributing to marine pollution by washing your face: microplastics in facial cleansers.” Marine Pollution Bulletin, vol. 58, no. 8, 2009, pp. 1225-1228.
Li Y. et al. “Trapping of plastics in semi-enclosed seas: insights from the Bohai Sea, China.” Marine Pollution Bulletin, vol. 137, 2018, pp. 509-517.
Vegter A.C. et al. “Global research priorities to mitigate plastic pollution impacts on marine wildlife.” Endangered Species Research, vol. 25, no. 3, 2014, pp. 225-247.
Fossi M.C. et al. “A review of plastic-associated pressures: cetaceans of the Mediterranean Sea and eastern Australian shearwaters as case studies.” Frontiers in Marine Science, vol. 5, 2018, p. 173.
Gouin T. “Toward an improved understanding of the ingestion and trophic transfer of microplastic particles: critical review and implications for future research.” Environmental Toxicology and Chemistry, vol. 39, no. 6, 2020, pp. 1119-1137.
Wu F. et al. “Accumulation of microplastics in typical commercial aquatic species: a case study at a productive aquaculture site in China.” Science of the Total Environment, vol. 708, 2020, p. 135432.
Edward J.K.P. et al. “Does monitoring lead to improved coral reef management.” Proceedings of the 12th International Coral Reef Symposium, 2012, pp. 9-13.
Balaji S. et al. “Coastal and marine biodiversity of Gulf of Mannar, southeastern India–a comprehensive updated species list.” Gulf of Mannar Biosphere Reserve Trust Publication, no. 22, 2012, p. 128.
Besley A. et al. “A standardized method for sampling and extraction methods for quantifying microplastics in beach sand.” Marine Pollution Bulletin, vol. 114, no. 1, 2017, pp. 77-83.
Löder M.G. and Gerdts G. “Methodology used for the detection and identification of microplastics—a critical appraisal.” Marine Anthropogenic Litter, 2015, pp. 201-227.
Gregory M.R. and Andrady A.L. “Plastics in the marine environment.” Plastics and the Environment, vol. 10, 2004, pp. 379-401.
Rodrigues M.O. et al. “Spatial and temporal distribution of microplastics in water and sediments of a freshwater system (Antuã River, Portugal).” Science of the Total Environment, vol. 633, 2018, pp. 1549-1559.
Vidyasakar A. et al. “Macrodebris and microplastic distribution in the beaches of Rameswaram Coral Island, Gulf of Mannar, southeast coast of India: a first report.” Marine Pollution Bulletin, vol. 137, 2018, pp. 610-616.
Patterson J.K. Assessment of microplastics in coral reef ecosystem of Gulf of Mannar, India. Suganthi Devadason Marine Research Institute, 2019.
James K. et al. “Seasonal variability in the distribution of microplastics in the coastal ecosystems and in some commercially important fishes of the Gulf of Mannar and Palk Bay, southeast coast of India.” Regional Studies in Marine Science, vol. 41, 2021, p. 101558.
Naidu S.A. et al. “Microplastics in the benthic invertebrates from the coastal waters of Kochi, southeastern Arabian Sea.” Environmental Geochemistry and Health, vol. 40, no. 4, 2018, pp. 1377-1383.
Karthik R. et al. “Microplastics along the beaches of southeast coast of India.” Science of the Total Environment, vol. 645, 2018, pp. 1388-1399.
Naidu S.A. “Preliminary study and first evidence of presence of microplastics and colorants in green mussel, Perna viridis (Linnaeus, 1758), from southeast coast of India.” Marine Pollution Bulletin, vol. 140, 2019, pp. 416-422.
Manickavasagam S. et al. “Assessment of marine microplastics in floating plastic debris using a fixed sampling device: the example of South Juhu creek, Mumbai coast, India.” Journal of Coastal Conservation, vol. 25, 2021, p. 20.
Tiwari M. et al. “Distribution and characterization of microplastics in beach sand from three different Indian coastal environments.” Marine Pollution Bulletin, vol. 140, 2019, pp. 262-273.
Davis E.K. and Raja S. “Sources and impact of microplastic pollution in Indian aquatic ecosystem: a review.” Current World Environment, 2020